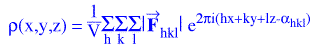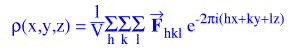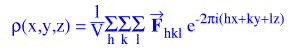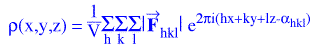The central difficulty in X-ray crystallography is that, in
order to use the electron density equation,
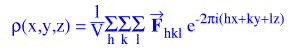 and, thus, “find” atoms we must know the structure factors, Fhkl, which are vector
quantities. Each structure factor has a magnitude and a direction (they can, of course, also be
considered to be waves with amplitudes and phases):
and, thus, “find” atoms we must know the structure factors, Fhkl, which are vector
quantities. Each structure factor has a magnitude and a direction (they can, of course, also be
considered to be waves with amplitudes and phases):
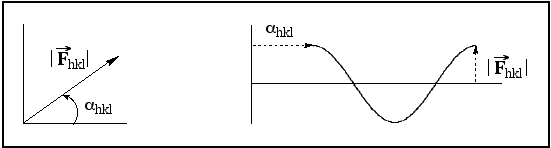
The quantity measured during the X-ray
experiment is the intensity of the reflection, which is related to the square of the structure factor.
Because the structure factor is obtained by taking the square root of the Intensity, all phase information is
lost. All that can be experimentally determined is the magnitude of Fhkl.
If the vector quantity, Fhkl, is expressed in exponential form:
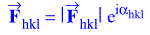 where the absolute value sign indicates the magnitude of the structure factor and
α is the phase angle of the structure factor. The electron density
equation, then, becomes:
where the absolute value sign indicates the magnitude of the structure factor and
α is the phase angle of the structure factor. The electron density
equation, then, becomes: